Navigating therapeutic sequencing in the metastatic castration-resistant prostate cancer patient journey | Prostate Cancer and Prostatic Diseases
Navigating therapeutic sequencing in the metastatic castration-resistant prostate cancer patient journey | Prostate Cancer and Prostatic Diseases
- Review Article
- Open access
- Published:
Prostate Cancer and Prostatic Diseases (2024)
McManus, H.D., Dorff, T., Morgans, A.K. et al. Navigating therapeutic sequencing in the metastatic castration-resistant prostate cancer patient journey. Prostate Cancer Prostatic Dis (2024). https://doi.org/10.1038/s41391-024-00906-z
Summary
Key points:
1. Multiple life-prolonging therapies with novel mechanisms of action have been approved for mCRPC in recent years, including ARPIs, taxane chemotherapy, PARP inhibitors, radiopharmaceuticals, and immunotherapies.
2. Optimal treatment selection should be individualized based on factors like prior treatment history, disease characteristics, genetic/molecular testing results, patient preferences, and accessibility.
3. Key considerations for sequencing include:
- Using therapies with different mechanisms of action to overcome resistance
- Genetic testing to identify actionable mutations (e.g. HRR mutations for PARP inhibitors)
- PSMA PET imaging to assess eligibility for PSMA-targeted therapy
- Pattern of metastatic spread and associated symptoms
- Patient-specific factors like comorbidities and preferences
4. Emerging strategies include using PSMA-targeted radioligand therapy earlier in treatment and in combinations.
5. Evidence gaps remain, particularly for sequencing after progression on ARPIs and for patients diagnosed with more sensitive imaging techniques.
6. Ongoing research is evaluating novel agents, combinations, and biomarkers to further optimize treatment selection and sequencing.
7. Multidisciplinary collaboration and staying up-to-date on new evidence is important for optimizing care as the treatment landscape continues to evolve rapidly.
The review provides an overview of current treatment options, considerations for sequencing, emerging strategies, and areas for future research in mCRPC. It emphasizes the importance of individualized treatment selection based on multiple patient and disease factors.
Q&A
1. Question: "What genetic testing do you recommend for my cancer, and how might the results affect my treatment options?"
Expected answer: Your oncologist should recommend both germline and somatic tumor genetic testing. They may explain that testing can identify mutations (like HRR gene alterations) that could make you eligible for specific treatments such as PARP inhibitors. The oncologist should be prepared to discuss the process, timeline, and potential implications of genetic testing results.
2. Question: "Should I undergo PSMA PET imaging, and how would that impact my treatment choices?"
Expected answer: The oncologist should explain that PSMA PET imaging can help determine if you're eligible for PSMA-targeted therapies like [177Lu]Lu-PSMA-617. They might discuss the availability of this imaging technique and how the results could influence treatment decisions, particularly if you've already undergone treatment with ARPIs and taxane-based chemotherapy.
3. Question: "Given my specific disease characteristics and treatment history, what do you recommend as the next line of therapy, and why?"
Expected answer: Your oncologist should provide a personalized recommendation based on factors like your previous treatments, the pattern of metastatic spread, symptoms, and any genetic testing results. They should explain the rationale behind their recommendation, discussing potential benefits and risks of the suggested treatment.
4. Question: "Are there any clinical trials that might be appropriate for me at this stage?"
Expected answer: The oncologist should be aware of ongoing clinical trials and be able to discuss any that might be suitable for your specific situation. They might mention trials investigating new therapies or combinations, particularly if standard treatment options have been exhausted.
5. Question: "How do you decide the sequence of treatments, and what factors do you consider when planning my long-term treatment strategy?"
Expected answer: The oncologist should explain their approach to treatment sequencing, considering factors like mechanisms of action of different therapies, your individual disease characteristics, and the goal of maximizing the benefit from all available treatment options over time.
6. Question: "Are there any new or emerging treatments that might become available in the near future that could be relevant for my case?"
Expected answer: The oncologist should be able to discuss emerging therapies or approaches that are showing promise in clinical trials, such as new combinations with PSMA-targeted therapies or novel immunotherapeutic strategies.
7. Question: "How will we monitor the effectiveness of my treatment, and how often will we reassess my treatment plan?"
Expected answer: Your oncologist should outline the methods they'll use to monitor your response to treatment (e.g., PSA levels, imaging scans) and discuss the frequency of follow-ups and reassessments. They should emphasize that the treatment plan can be adjusted based on your response and any new developments in your condition or available therapies.
Remember, these are complex topics, and it's perfectly acceptable to ask for clarification or simplification if any explanations are unclear. Your oncologist should be willing to take the time to ensure you understand your condition and treatment options.
Navigating therapeutic sequencing in the metastatic castration-resistant prostate cancer patient journey
Abstract
Background
Novel therapies for metastatic castration-resistant prostate cancer (mCRPC) have improved patient outcomes. However, there is uncertainty on the optimal selection of therapeutic agents for subsequent lines of therapy.
Methods
We conducted a comprehensive review of published evidence from pivotal clinical trials and recent guidelines for the treatment of mCRPC. We further identify gaps in knowledge and areas for future research.
Results
Key considerations to help guide treatment selection for patients with mCRPC include personal treatment history, individual clinical characteristics, symptoms, prognosis, availability of clinical trials, and other patient-specific factors. Genetic testing and prostate-specific membrane antigen-targeted imaging are important tools to evaluate candidacy for newer therapeutic options such as poly (ADP-ribose) polymerase inhibitors, alone or in combination with androgen receptor pathway inhibitors, and [177Lu]Lu-PSMA-617.
Conclusion
This article provides an overview of the evolving treatment landscape of mCRPC, discussing guideline-recommended treatment options and data from key clinical trials, while highlighting ongoing trials that may impact the future treatment landscape. Recommendations for optimal treatment sequencing based on individual patient factors are provided.
Similar content being viewed by others
Introduction
In the United States (US), prostate cancer (PC) is the most frequently diagnosed cancer in men, representing 14% of all new cancer cases [1]. Approximately 8% of patients with PC have metastatic disease at diagnosis, and rates of de novo metastatic disease are increasing [2]. Initial treatment for metastatic or biochemically recurrent PC typically includes androgen deprivation therapy (ADT), often combined with an androgen receptor pathway inhibitor (ARPI) ± docetaxel [3,4,5]. Despite treatment, patients often experience disease progression while maintaining castrate levels of serum testosterone, either with metastases (metastatic castration-resistant PC [CRPC]; mCRPC) or without metastases (non-metastatic/M0 CRPC) [6].
Numerous life-prolonging therapies with novel mechanisms of action (MoAs) have been approved for the treatment of mCRPC [7,8,9,10,11,12,13,14,15,16,17]. In the US, these include taxane-based chemotherapy (docetaxel and cabazitaxel), ARPIs (enzalutamide and abiraterone acetate), radiopharmaceuticals (radium-223 and prostate-specific membrane antigen [PSMA]-targeted lutetium Lu-177 vipivotide tetraxetan, i.e., [177Lu]Lu-PSMA-617), poly-ADP ribose polymerase (PARP) inhibitors (olaparib, rucaparib, niraparib, and talazoparib), and immunotherapies (sipuleucel-T and pembrolizumab) (Table 1) [7,8,9,10,11,12,13,14,15,16,17,18]. Additionally, metastasis-directed therapy, such as stereotactic body radiation therapy (SBRT), can be considered in select cases of oligoprogressive disease [19]. Although improvements in clinical outcomes have been observed with novel therapies [20,21,22,23,24,25,26,27,28,29,30,31], their approval has resulted in uncertainties on optimal treatment sequencing, and utilization of these treatments is limited, demonstrating the need for improved real-world treatment practices.
Therapies previously reserved for mCRPC have been expanded to earlier PC stages, including metastatic hormone-sensitive PC (mHSPC) and M0 CRPC, impacting treatment selection in mCRPC [3]. As ARPIs are now a standard of care (SOC) for both mHSPC and M0 CRPC, many patients will have experienced disease progression on both ADT and an ARPI when diagnosed with mCRPC [3]. The further adoption of triplet therapy (first-line docetaxel plus ARPI plus ADT) as a SOC for patients with mHSPC means that many patients will be exposed to both an ARPI and docetaxel before developing castration-resistant prostate cancer (CRPC) [32]. To overcome neoplastic biologic resistance and maximize clinical efficacy, therapies with a different MoA from previously utilized treatments should be selected [33, 34]. Therefore, accessibility and applicability of therapies within the pre-mCRPC space influence the availability and efficacy of treatments for mCRPC. In real-world practice, many patients with mHSPC still receive ADT monotherapy with rates of treatment intensification with docetaxel or ARPI ranging from 9.3% to 38.1% in a recent systematic review [35].
The importance of treatment sequencing is notable in real-world data examining mCRPC treatment patterns, as median OS lessens following each line of therapy [36]. Moreover, approximately half of patients treated for mCRPC receive ≥1 line of therapy [36]. In addition to clinical and patient-specific factors, accessibility of and information from genetic testing and molecular imaging expand the available information for decision-making.
This article reviews the evolving treatment landscape for mCRPC and provides recommendations for optimal treatment sequencing.
Treatment recommendations and practice
Various guidelines provide treatment recommendations for mCRPC, including the American Society of Clinical Oncology, American Urological Association, Society of Urologic Oncology, National Comprehensive Cancer Network® (NCCN®), European Society for Medical Oncology, and European Association of Urology (Table 2) [3, 37,38,39,40]. Optimal therapy selection should be individualized, based on critical patient and disease factors.
General principles for treatment selection
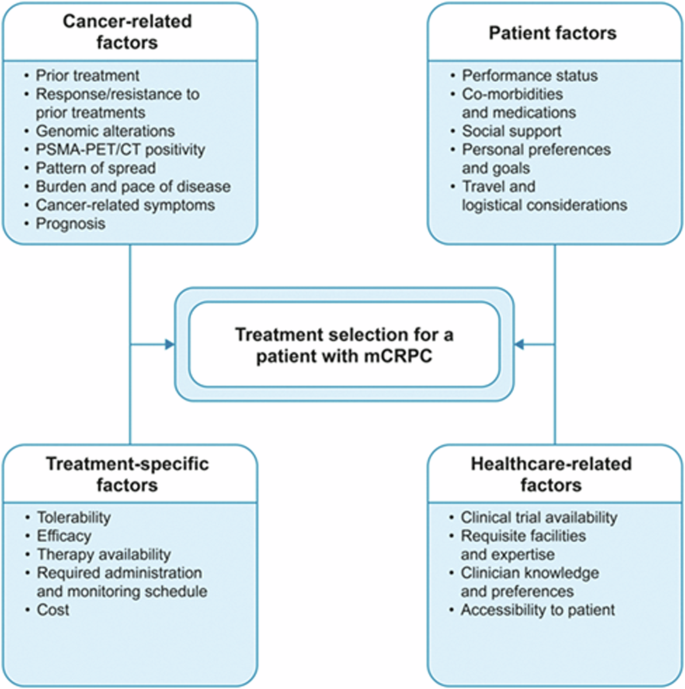
Key considerations for treatment selection include personal treatment history, individual disease characteristics, prognosis, and patient-specific factors (Fig. 1). Generally, selecting treatments with a novel MoA reduces cross-resistance and improves the likelihood of clinical benefit, with the exception of taxane-based chemotherapy [41]. Despite frequent use in real-world settings, switching to a second ARPI after disease progression on a first-line ARPI has limited efficacy [33, 34]. The effectiveness of re-treatment with docetaxel, following docetaxel- and ARPI-based treatment combinations in the mHSPC setting, has also been challenged, but may be dependent on the timing of progression after initial docetaxel treatment [42].
In tumor characteristics should be considered/tumor characteristics should be considered.
CT computed tomography, mCRPC metastatic castration-resistant prostate cancer, PET positron emission tomography, PSMA prostate-specific membrane antigen. *Please note that not all factors presented in this figure were within the scope of this review.
Pattern of disease spread and associated symptoms
The pattern of metastatic spread can provide insight into disease behavior, influencing treatment selection [43, 44]. Visceral metastases, specifically liver metastases, can be predictive of more aggressive disease biology and shorter survival observed with ARPIs and chemotherapy, vs patients without liver metastases [43, 44]. Although often classified together as visceral metastases, patients with lung metastases have a more favorable prognosis vs patients with liver metastases [43]. In patients with liver metastases, treatment with microtubule inhibitor chemotherapy, either docetaxel (if not received previously) or cabazitaxel, is generally preferred [3]. A phase 2 trial (NCT02254785) including patients with ARPI-naïve mCRPC and poor prognostic features (including liver metastases) demonstrated a higher clinical benefit rate with cabazitaxel vs physician’s choice of enzalutamide or abiraterone [45]. Radium-223 (a bone-directed alpha emitter) can be considered for patients with symptomatic, bone-predominant metastatic disease without visceral metastases [23]. Of note, except for concurrent ADT and bone-targeting agents, radium-223 is not recommended for use as a combination therapy, given the increased fracture rates seen when administered with abiraterone [3]. Alternatively, the active cellular immunotherapy sipuleucel-T can be considered for patients with asymptomatic to minimally symptomatic disease without visceral metastases [24]. Although the pattern of metastasis may determine patient eligibility [23, 24, 43, 44], decisions on when to incorporate these therapies must consider all available options.
In patients with disease oligoprogression on systemic therapy, metastasis-directed therapy (radiotherapy delivery to metastatic lesions) can be considered, but there are limited data on this strategy in mCRPC [19, 30, 46, 47]. In the phase 2 ARTO trial, abiraterone plus SBRT improved prostate-specific antigen (PSA)-based outcomes and progression-free survival (PFS) vs abiraterone alone in patients with oligometastatic CRPC [19]. In the single-arm phase 2 TRAP trial, patients with oligoprogressive CRPC (with ≤2 progressive lesions that developed while on ARPI treatment) received SBRT [47]. The median PFS was 6.4 months (95% confidence interval [CI] 5.9–12.8 months), which exceeded the investigators hypothesized median PFS of 4 months [47]. The investigators commented that over 40% of patients remained progression-free at 12 months, and that treatment did not impact patient quality of life [47]. Several retrospective series have evaluated the outcomes of patients with oligoprogressive CRPC lesions treated with metastasis-directed therapy, demonstrating improved PFS and delays in systemic therapy change [30, 46]. In patients with limited sites of disease progression, treatment with metastasis-directed therapy can be considered to delay the need to switch systemic therapy; however, larger studies are required.
Prognosis
The pattern of metastasis in patients with mCRPC has been established as a prognostic factor [43]. In addition to visceral metastases, multiple clinical factors have demonstrated an association with varying prognoses, including PSA, alkaline phosphatase, hemoglobin, albumin, and lactate dehydrogenase [48,49,50]. Several multivariable models are available that incorporate these factors to help inform prognoses for patients with mCRPC [48,49,50], and prognostic knowledge from these models may inform treatment decision-making.
Genotypic and phenotypic testing
Approximately 11% of patients with metastatic PC may harbor a germline mutation in DNA-repair genes [51]. However, PCs can possess a somatic mutation not present in the germline [52]. In one study, ~50% of somatic mutations in homologous recombination repair (HRR) genes were also identified on germline testing [53]. Subsequently, the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Version 4.2024 recommend both germline and somatic tumor genetic testing for all patients with metastatic PC [3]. However, real-world rates of genetic testing remain low [54]. A study assessing data from 2013 to 2019 demonstrated that only 13% (674/5213) of patients with mCRPC had undergone any genetic testing [54]. Genetic testing of patients with mCRPC should be prioritized at treatment initiation to ensure a complete assessment of potential treatment options.
Increased use of genetic testing can expand available therapy options in the mCRPC setting, including PARP and immune checkpoint inhibitors. Furthermore, early genetic testing may allow for clinical trial enrollment based on specific mutations, which could improve our understanding of individual disease biology, and also provide opportunities to identify relatives with high-risk genotypes for cancer screening. Somatic tumor testing is recommended for all patients with metastatic PC [3]. When a metastatic biopsy is not feasible, a plasma circulating tumor DNA assay can be utilized [55]. Clonal hematopoiesis of indeterminate potential, the acquisition of somatic mutations in hematopoietic cells and their clonal expansion in the absence of cancer, can be a potential source of false positives when utilizing circulating tumor DNA assays to identify tumor-derived alterations in PC [56]. Repeat testing can be considered at times of disease progression to allow the identification of targetable alterations developed later in disease evolution.
HRR gene alterations
Genetic testing can provide therapeutically actionable insights through the identification of germline or somatic mutations. PARP inhibitors, which inhibit the repair of DNA single-stranded breaks and induce DNA double-strand breaks, are one treatment option for patients with mCRPC with a HRR gene mutation [57, 58]. In the US, two PARP inhibitors (olaparib and rucaparib) are approved as monotherapy for mCRPC in patients with HRR deficiency. Olaparib is approved for use after an ARPI, before or after docetaxel in patients with an HRR gene alteration [14]. Rucaparib is approved for patients with mCRPC harboring a BRCA1 or BRCA2 alteration after treatment with an ARPI and taxane-based chemotherapy [3]. The timing of treatment with PARP inhibitors, before or after taxane-based chemotherapy, should be considered in addition to HRR alterations and patient preference. In the phase 3 randomized trials PROfound (NCT02987543), and TRITON-3 (NCT02975934), treatment with PARP inhibitors was favored in both chemotherapy-naïve patients and those who had received prior taxane-based chemotherapy [57, 58]. In an exploratory analysis of a subgroup of TRITON-3 evaluating patients with BRCA alterations, the median PFS with rucaparib (11.2 months) was longer than with docetaxel (6.4 months; p < 0.001) [57]. Based on these results, PARP inhibitors represent a potential treatment option both before and after docetaxel in patients with BRCA-mutated (BRCAm) mCRPC. Additionally, while olaparib is approved for patients with a spectrum of HRR mutations, patients with BRCA2 mutations appear to derive the greatest benefit [58]. Thus, the use of PARP inhibitors could be prioritized earlier in the treatment sequence for patients with BRCA mutations vs less susceptible HRR gene alterations [58].
PARP inhibitors, in combination with ARPIs, have been approved in the US for patients with mCRPC and select HRR gene mutations [14,15,16, 18]. However, preclinical models suggested that PARP inhibitors combined with ARPIs would have a synergistic antitumor effect irrespective of HRR alteration [59]. Thus, the phase 3 trials leading to FDA approval of these PARP inhibitor/ARPI combinations included patients with HRR mutations and patients with mCRPC irrespective of HRR mutation status [26,27,28, 58, 60]. In the phase 3 PROpel trial (NCT03732820), the combination of abiraterone plus olaparib as first-line treatment resulted in improved PFS compared with abiraterone plus placebo (median: 24.8 vs 16.6 months; HR 0.66; 95% CI 0.54–0.81; p < 0.001) irrespective of HRR status [60]. In a post hoc exploratory analysis, the radiographic PFS (rPFS) benefit of olaparib plus abiraterone was most pronounced in the HRR-mutated (HRRm) (HR 0.50; 95% CI 0.34–0.73) and BRCAm subgroups (HR 0.23; 95% CI 0.12–0.43) [60, 61]. The final OS analysis from PROpel demonstrated a trend toward improved OS, with a median OS of 42.1 months in patients treated with abiraterone plus olaparib vs 34.7 months in patients treated with abiraterone plus placebo (HR 0.81 95% CI 0.67–1.00, p = 0.0544) irrespective of HRR status [26]. In a post hoc exploratory assessment of OS, the HR was 0.66 (95% CI 0.45–0.95) in the HRRm subgroup and 0.29 (95% CI 0.14–0.56) in the BRCAm subgroup [26].
Similarly, in the phase 3 study TALAPRO-2 (NCT03395197), talazoparib plus enzalutamide significantly improved rPFS compared with enzalutamide plus placebo (median: not reached vs 21.9 months, respectively; HR 0.63; 95% CI 0.51–0.78; p < 0.001), regardless of HRR gene mutation status, as well as in the HRRm group (median: not reached vs 13.8 months, respectively, HR 0.45; 95% CI 0.33–0.61; p < 0.001). OS remains immature in this trial [62]. Contrastingly, in the phase 3 trial MAGNITUDE (NCT03748641), a significant rPFS benefit was seen for patients in the HRRm cohort treated with niraparib plus abiraterone, compared with placebo plus abiraterone for first-line mCRPC (median: 16.5 vs 13.7 months, respectively; HR 0.73; 95% CI 0.56–0.96; p = 0.022) but not in the non-HRRm cohort (HR 1.09; 95% CI 0.75–1.57; p = 0.66) [29]. Final OS analysis of patients with BRCAm mCRPC demonstrated improved OS for patients receiving niraparib plus abiraterone after pre-specified adjustment for imbalances in baseline characteristics (HR 0.66; 95% CI 0.46–0.95; p = 0.02) [63].
In a meta-analysis of the PROpel, TALAPRO-2, and MAGNITUDE trials investigating PARP inhibitor plus ARPI combinations vs placebo plus ARPI in first-line mCRPC, the pooled HRs were 0.62 (95% CI 0.53–0.72) for rPFS, and 0.84 (95% CI 0.72–0.98) for OS [4]. This suggests a benefit from the combination in HRR-unselected mCRPC, as PROpel and TALAPRO-2 assessed rPFS independent of HRR status [4]. At present, several PARP inhibitor/ARPI combinations are FDA-approved for select patients with HRR-altered mCRPC, including olaparib plus abiraterone and prednisone/prednisolone (BRCA1/2-mutated mCRPC), talazoparib plus enzalutamide (HRRm mCRPC), and niraparib plus abiraterone and prednisone (BRCAm mCRPC) [14, 16, 18]. Of note, the FDA and European Medicines Agency (EMA) labels for the combinations of olaparib plus abiraterone and talazoparib plus enzalutamide differ. In the US, the combination of olaparib or niraparib plus abiraterone and prednisone/prednisolone is approved for adult patients with mCRPC and deleterious or suspected deleterious BRCA mutations, whereas talazoparib plus enzalutamide is approved for adult patients with mCRPC and select HRR mutations (BRCA1, BRCA2, ATM, ATR, CDK12, CHEK2, FANCA, MLH1, MRE11A, NBN, PALB2, and RAD51C) [14, 16]. Contrastingly, the EMA labels for both combinations of olaparib plus abiraterone and prednisone/prednisolone, and talazoparib plus enzalutamide, include patients with mCRPC for whom chemotherapy is not clinically indicated, regardless of HRRm status [64, 65]. When choosing between the available combinations, multiple factors, including specific mutations, toxicity differences (e.g., rates of grade ≥3 anemia), data maturity (PROpel is the only study with OS benefit demonstrated to date), and accessibility should be considered [26, 28, 60, 63]. Of note, patients with mCRPC who developed resistance on a prior ARPI were not represented in PROpel, MAGNITUDE, or TALAPRO-2, so the benefit of these combinations in these patients is not known [28, 60, 63].
Individual HRR gene mutations demonstrate heterogeneous sensitivity to PARP inhibitors. In an exploratory pooled analysis from the FDA including three PARP inhibitor plus ARPI trials and three PARP inhibitor monotherapy trials in patients with mCRPC, the benefit of PARP inhibitor treatment appeared greatest for patients with BRCA1 and BRCA2 mutations as well as CDK12 and PALB2 mutations, with a lack of treatment effect demonstrated in patients with CHEK2 or ATM mutations in these studies [66].
MSI-H/dMMR or TMB-high
Deficient mismatch repair (dMMR) prevents the reversal of DNA base mismatches that may occur during cellular replication, resulting in high microsatellite instability (MSI-H) and a hypermutator phenotype associated with chemotherapy resistance and immunotherapy sensitivity [67]. Although dMMR and MSI-H are relatively uncommon in PC (<9%), they can be therapeutically meaningful for patients [67, 68]. Pembrolizumab, an anti-programmed cell death protein-1 (PD-1) antibody, is a treatment option for patients whose tumors have dMMR/MSI-H or a high tumor mutational burden (TMB-H) [≥10 mutations/megabase] [3]. Pembrolizumab received tumor-agnostic FDA approval for the treatment of advanced solid tumors (including PC) that are MSI-H/dMMR or TMB-H when there are no satisfactory alternative treatments [69]. However, data remain limited to small retrospective trials [31, 68]. In a retrospective case series of 11 patients with MSI-H/dMMR CRPC receiving anti-PD-1/programmed death-ligand 1 therapy, 6 (54.5%) patients had a ≥50% PSA decline, and 4 (36.4%) demonstrated radiographic responses [68]. In another retrospective series of 27 patients with dMMR/MSI-H metastatic PC, 8/17 (53.0%) pembrolizumab-treated patients experienced PSA responses ≥50%, of whom 7 (87.5%) remained on treatment without progression at 12 months median follow-up (range: 3–20 months) [31]. Therefore, treatment with pembrolizumab can be considered prior to docetaxel for patients with dMMR/MSI-H or TMB-H mCRPC, generally after an ARPI. Dostarlimab, another anti-PD-1 antibody, has histology-agnostic FDA approval for dMMR tumors; however, data are limited in patients with PC [70].
PSMA-positive disease
For patients with PSMA-positive metastatic disease who received prior treatment with ≥1 ARPI and taxane-based chemotherapy, treatment with the PSMA-targeted radioligand therapy (RLT) [177Lu]Lu-PSMA-617 is an option [13]. Patients must undergo PSMA PET imaging to assess eligibility, including ≥1 PSMA-positive metastatic lesion and no PSMA-negative lesions [3, 13]. [68Ga]Ga-PSMA-11 is currently the only radiopharmaceutical approved by the FDA to assess eligibility for [177Lu]Lu-PSMA-617 treatment; however, according to NCCN Guidelines®, PSMA PET imaging can also be performed using [18F]F-DCFPyL, or [18F]F-rhPSMA-7.3 [3]. In the phase 3 VISION trial (NCT03511664), treatment with [177Lu]Lu-PSMA-617 plus best SOC (BSOC) prolonged rPFS and OS in patients with PSMA-positive mCRPC who had previously received ≥1 ARPI and 1–2 taxane-based chemotherapy regimens [25]. In the randomized phase 2 TheraP trial (NCT03392428) treatment with [177Lu]Lu-PSMA-617 compared with cabazitaxel had fewer grade ≥3 adverse events, improved patient-reported outcomes, and yielded similar OS benefit, which may support the use of [177Lu]Lu-PSMA-617 prior to cabazitaxel [71]. [177Lu]Lu-PSMA-617 can also be considered after docetaxel and cabazitaxel, as [177Lu]Lu-PSMA-617 demonstrated efficacy in patients who had received 1–2 taxane-based chemotherapy regimens in the VISION trial [25].
There is emerging evidence that higher baseline levels of uptake (standardized uptake value mean [SUVmean]) and lack of liver metastases on PSMA PET correlate with improved outcomes following treatment with [177Lu]Lu-PSMA-617 [72]. However, treatment with [177Lu]Lu-PSMA-617 plus BSOC prolonged rPFS and OS in patients with PSMA-positive mCRPC, inclusive of eligible patients with lower SUVmean [25]. Additional evidence is required to validate this association before these measures can be applied in clinical practice [72]. Some patients will have PSMA-negative disease and will not be candidates for PSMA-targeted therapy; in the VISION trial, 12.6% of patients did not meet eligibility criteria based on PSMA imaging [25]. In the updated survival analysis of TheraP, no OS differences were observed between [177Lu]Lu-PSMA-617 and cabazitaxel (19.1 vs 19.6 months, respectively) irrespective of baseline PSMA PET SUVmean, and patients with high SUVmean (>10) had improved outcomes irrespective of therapy [73], suggesting that PSMA SUVmean is prognostic and not predictive, and that the outcomes of mCRPC patients are similar regardless of [177Lu]Lu-PSMA-617 or taxane-based chemotherapy treatment sequencing.
Tumor-agnostic drugs
In addition to the tumor-agnostic approvals for pembrolizumab and dostarlimab, several additional treatments have FDA approvals for histology-agnostic indications, including larotrectinib and entrectinib (for tumors harboring neurotrophic tyrosine receptor kinase [NTRK] fusions), dabrafenib plus trametinib (for BRAF V600E-mutated tumors), and selpercatinib (for rearrangements during transfection [RET] gene fusions) [74,75,76,77]. Experience with these agents in PC is limited outside of pembrolizumab.
Patient-specific factors and preferences
Patient-specific factors, including co-morbidities and performance status, are important considerations for treatment selection. Frail patients with multiple co-morbidities may not be candidates for taxane-based chemotherapy but could receive therapies associated with lower adverse event incidences [78, 79]. Alternative dosing strategies, which improve tolerability while preserving efficacy, can be considered for both docetaxel and cabazitaxel in this population [78, 79]. Individual circumstances (e.g., the ability to travel) and cost may influence access and feasibility of certain treatments. Importantly, patient goals and preferences should be key considerations during treatment selection. Establishing an understanding of the risk tolerance and priorities of patients and their caregivers can also aid decision-making.
Clinic-related factors
Factors regarding the clinical setting in which patients are treated influence therapy selection. When available, clinical trials should be considered for all patients with mCRPC [3], but the availability of suitable trials varies depending on treatment setting. An oncology provider’s specialty, and ability to collaborate in multidisciplinary care, may influence therapy availability. Clinic-specific logistical considerations (e.g., availability of specific facilities) and reimbursement issues also influence treatment decisions. Utilization and timing of radiopharmaceuticals may be influenced by access to a center with the required infrastructure, multidisciplinary teams, and investment to provide these therapies.
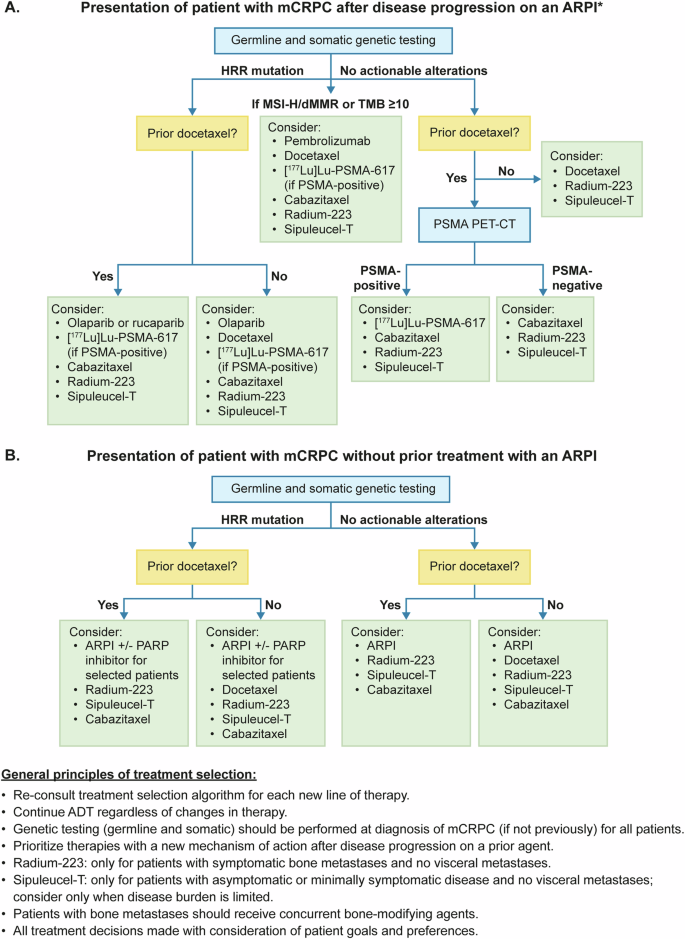
Often, there is no definitive optimal treatment option for a patient with mCRPC. Reaching a therapeutic decision requires consideration of many factors and thoughtful discussion with patients regarding their options. The general principles reviewed above are summarized in Fig. 2 and can be used as a framework to guide decision-making.
A Treatment selection algorithm for mCRPC in patients with progressive disease following treatment with an ARPI. B Treatment selection algorithm for mCRPC in patients without prior ARPI. ADT androgen deprivation therapy, ARPI androgen receptor pathway inhibitor, CRPC castration-resistant prostate cancer, CT computed tomography, HRR homologous recombination repair, Lu lutetium, mCRPC metastatic castration-resistant prostate cancer, MSI-H microsatellite instability-high, PARP poly (ADP-ribose) polymerase, PET positron emission tomography, PSMA prostate-specific membrane antigen, TMB tumor mutational burden. *If aggressive mCRPC disease features (e.g., liver metastases, short time to CRPC, multiple lytic bone lesions), prioritize taxane chemotherapy (consider adding carboplatin) if a candidate.
Future therapeutic strategies
New therapeutic strategies in the treatment of mCRPC are currently being investigated. New therapeutic options will become available as results are published, further changing the sequencing of therapies. Use of PSMA-targeted RLT, such as [177Lu]Lu-PSMA-617, earlier and in combinations, is a key focus of ongoing trials.
PSMA-targeted RLT
Interest in [177Lu]Lu-PSMA-617 has increased following its approval for the treatment of patients with PSMA-positive mCRPC post-treatment with ARPI and taxane-based chemotherapy [13]. As patients may now receive treatment with an ARPI and taxane-based chemotherapy in the mHSPC setting [3], [177Lu]Lu-PSMA-617 is increasingly being used as a first-line treatment for mCRPC [25, 80]. Earlier use of [177Lu]Lu-PSMA-617 in patients with PSMA-positive mCRPC is being evaluated in the phase 3 PSMAfore (NCT04689828) trial in patients who have not received taxane-based chemotherapy [81, 82]. PSMAfore met its primary endpoint, demonstrating significantly improved rPFS in patients randomized to [177Lu]Lu-PSMA-617 vs an ARPI change (HR 0.41; 95% CI 0.29–0.56; p < 0.0001), in addition to improved ORR ([177Lu]Lu-PSMA-617 vs ARPI change: 50.7% vs 14.9%) and time to worsening of health-related quality of life (FACT-P total score, HR 0.59; 95% CI 0.47–0.72) [81, 82]. At the third interim OS analysis (with 73% of targeted events), the pre-specified crossover-adjusted OS analysis demonstrated a HR of 0.98 (95% CI 0.76–1.27) [83]. Unadjusted intention-to-treat OS analysis demonstrated a HR of 0.98 (95% CI 0.75–1.28) but was confounded by 78% crossover after progression on ARPI change [83]. Significant delays in time to worsening in health-related quality of life and pain were observed with PSMA RLT vs ARPI change at second interim OS analysis [83]. These data support the efficacy of PSMA-RLT in patients with mCRPC prior to docetaxel therapy, following progression on an ARPI. The value of PSMA-RLT in addition to an ARPI was assessed in the ENZA-p (NCT04419402) trial, which demonstrated significantly improved PSA-based outcomes in high-risk, first-line mCRPC vs enzalutamide alone, suggesting future larger studies should evaluate combination approaches [84]. The PSMAddition (NCT04720157) trial is assessing the efficacy and safety of [177Lu]Lu-PSMA-617 in combination with ARPI in patients with mHSPC [85].
Although [177Lu]Lu-PSMA-617 is currently the only FDA-approved PSMA-targeted RLT, other strategies are under investigation, including [177Lu]Lu-PNT2022, I-131-1095 (small-molecule PSMA-targeted 131I-based RLT) and strategies utilizing alpha-emitters (e.g., actinium-225; [225Ac]Ac-J591) [86, 87]. [177Lu]Lu-PNT2022 is being investigated in two phase 3 trials, SPLASH (NCT04647526) and ECLIPSE (NCT05204927), comparing it with abiraterone or enzalutamide in patients with mCRPC who have received one prior ARPI and no prior chemotherapy [88, 89]. No data from these trials have been published, aside from the results of a 27-patient lead-in phase from SPLASH [88, 89]. I-131-1095 radiotherapy (in combination with enzalutamide) is being evaluated in the randomized phase 2 ARROW trial (NCT03939689), comparing it with enzalutamide in patients with mCRPC who experienced disease progression on abiraterone; no data have been published [87].
Multiple ongoing trials are investigating PSMA-RLT in combination with other therapies for mCRPC, with the goal of improving disease control [86, 90, 91]. [177Lu]Lu-PSMA-617 is being assessed in combination with: olaparib (NCT03874884), cabazitaxel (NCT05340374), ipilimumab plus nivolumab (NCT05150236), pembrolizumab (NCT03658447 and NCT05766371), abemaciclib (NCT05113537), and cabozantinib (NCT05613894), while [177Lu]Lu-PSMA-I&T is being assessed in combination with radium-223 (NCT05383079) [86, 90, 91].
Evidence gaps and future directions
Despite numerous therapeutic strategies, evidence gaps regarding optimal treatment selection remain. These are particularly substantial in the post-ARPI space, as pivotal trials of many current treatments were performed in patients who had not received a prior ARPI [22, 23, 30, 92]. Additionally, pivotal trials of current treatments utilized conventional imaging to determine metastatic disease [22, 23, 30, 92]. The use of more sensitive PSMA PET imaging, now routine in clinical practice, may identify metastases in patients that previously would have been classified as having M0 CRPC with conventional imaging [93]. This earlier detection of mCRPC may allow treatment earlier in the disease course.
Novel agents with different MoAs, including androgen receptor degraders, and immunotherapeutic strategies, such as bispecific T-cell engagers and chimeric antigen receptor T-cell therapies [94], are being evaluated in phase 1/2 trials. With the exception of PD-1 blockade in patients with mCRPC harboring MSI-H/dMMR or TMB-H tumors, PD-1 blockade in PC has provided limited survival benefit, and ongoing strategies to understand immune evasion and increase the effectiveness of checkpoint blockade in mCRPC are needed [31, 68]. The combination of cabozantinib, a small-molecule receptor tyrosine kinase inhibitor, plus atezolizumab, an anti-PD-L1 antibody, is being studied in the phase 3 CONTACT-02 trial, demonstrating a statistically significant improvement in PFS compared with a second ARPI at the primary analysis [95].
Predictive biomarkers and the development of clinical–genomic models to help guide optimal therapy selection represent key future study areas. Biomarkers of poor outcomes and ARPI resistance, including aggressive variant signatures (e.g., PTEN), and biomarkers predictive of improved outcomes with ARPI treatment (e.g., SPOP) are emerging and may soon become actionable [96, 97]. Understanding mechanisms of resistance to androgen receptor signaling inhibition, and how to target these pathways, are important areas for ongoing discovery [98]. While not covered here, PC with neuroendocrine/small cell features represents an important area of needed investigation and additional treatment options.
It is important to note that this review focuses on practice patterns in the US, which is a limitation of this article. Further work that describes different treatment sequences globally is needed, given the variation in practice patterns between different countries and regions [99].
Conclusion
Our understanding of mCRPC has improved dramatically due to advances in genetic testing, molecular imaging, and further evidence-based learnings. Consequently, multiple treatment options now exist for mCRPC, contributing to improved prognoses for patients. However, these new developments make individual treatment decision-making more complex, thus uncertainties regarding optimal treatment choices and sequencing in clinical practice remain. Continued multidisciplinary collaboration and attention to regular educational updates are important.
References
National Cancer Institute: Cancer stat facts: common cancer sites. 2024. https://seer.cancer.gov/statfacts/html/common.html.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7–33.
National Comprehensive Cancer Network. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Prostate Cancer V.4.2024, © National Comprehensive Cancer Network, Inc. 2024. All rights reserved. Available at NCCN.org. 2024.
Messina C, Giunta EF, Signori A, Rebuzzi SE, Banna GL, Maniam A, et al. Combining PARP inhibitors and androgen receptor signalling inhibitors in metastatic prostate cancer: a quantitative synthesis and meta-analysis. Eur Urol Oncol. 2023;7:179–88.
Chen J, Ni Y, Sun G, Liao B, Zhang X, Zhao J, et al. Comparison of current systemic combination therapies for metastatic hormone-sensitive prostate cancer and selection of candidates for optimal treatment: a systematic review and Bayesian network meta-analysis. Front Oncol. 2020;10:519388.
Scher HI, Halabi S, Tannock I, Morris M, Sternberg CN, Carducci MA, et al. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical Trials Working Group. J Clin Oncol. 2008;26:1148–59.
Janssen Biotech Inc: ZYTIGA (abiraterone acetate), tablets for oral administration: highlights of prescribing information. 2011. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/202379lbl.pdf.
Astellas Inc.: XTANDI (enzalutamide) capsules/tablets, for oral use: highlights of prescribing information. 2012. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/203415s019,213674s006lbl.pdf.
Sanofi Aventis US: TAXOTERE (docetaxel) injection, for intravenous use: highlights of prescribing information. 1996. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/020449s086lbl.pdf.
Sanofi Aventis US: JEVTANA (cabazitaxel) Injection, 60 mg/1.5 mL, for intravenous infusion only: highlights of prescribing information. 2010. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/201023s024lbl.pdf.
Bayer Healthcare: XOFIGO (radium Ra 223 dichloride) injection, for intravenous use: highlights of prescribing information. 2013. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/203971s016lbl.pdf.
Dendreon Corporation: PROVENGE® (sipuleucel-T) suspension for intravenous infusion: highlights of prescribing information. 2010. https://www.fda.gov/media/78511/download.
Novartis Pharmaceuticals Corporation: PLUVICTO (lutetium Lu 177 vipivotide tetraxetan) injection, for intravenous use: highlights of prescribing information. 2022. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/215833s000lbl.pdf.
AstraZeneca: LYNPARZA (olaparib) tablets, for oral use: highlights of prescribing information. 2014. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/208558s025lbl.pdf.
ZR Pharma: RUBRACA (rucaparib) tablets, for oral use: highlights of prescribing information. 2016. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/209115s013lbl.pdf.
Janssen Biotech: AKEEGA (niraparib and abiraterone acetate) tablets, for oral use. 2023. https://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/AKEEGA-pi.pdf.
Merck Sharp & Dohme: KEYTRUDA® (pembrolizumab) injection, for intravenous use: highlights of prescribing information. 2014. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/209115s013lbl.pdf.
Pfizer: TALZENNA (talazoparib) capsules, for oral use: prescribing information. 2018. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/211651s010lbl.pdf.
Francolini G, Gaetano Allegra A, Detti B, Di Cataldo V, Caini S, Bruni A, et al. Stereotactic body radiation therapy and abiraterone acetate for patients affected by oligometastatic castrate-resistant prostate cancer: a randomized phase II trial (ARTO). J Clin Oncol. 2023;41:5561–8. Jco2300985
de Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, Chu L, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995–2005.
Scher HI, Fizazi K, Saad F, Taplin ME, Sternberg CN, Miller K, et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187–97.
de Bono JS, Oudard S, Ozguroglu M, Hansen S, Machiels JP, Kocak I, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376:1147–54.
Parker C, Nilsson S, Heinrich D, Helle SI, O’Sullivan JM, Fossa SD, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369:213–23.
Kantoff PW, Higano CS, Shore ND, Berger ER, Small EJ, Penson DF, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411–22.
Sartor O, de Bono J, Chi KN, Fizazi K, Herrmann K, Rahbar K, et al. Lutetium-177-PSMA-617 for metastatic castration-resistant prostate cancer. N Eng J Med. 2021;385:1091–103.
Saad F, Clarke NW, Oya M, Shore N, Procopio G, Guedes JD, et al. Olaparib plus abiraterone versus placebo plus abiraterone in metastatic castration-resistant prostate cancer (PROpel): final prespecified overall survival results of a randomised, double-blind, phase 3 trial. Lancet Oncol. 2023;24:1094–108.
Abida W, Patnaik A, Campbell D, Shapiro J, Bryce AH, McDermott R, et al. Rucaparib in men with metastatic castration-resistant prostate cancer harboring a BRCA1 or BRCA2 gene alteration. J Clin Oncol. 2020;38:3763–72.
Agarwal N, Azad AA, Carles J, Fay AP, Matsubara N, Heinrich D, et al. Talazoparib plus enzalutamide in men with first-line metastatic castration-resistant prostate cancer (TALAPRO-2): a randomised, placebo-controlled, phase 3 trial. Lancet. 2023;402:291–303.
Chi KN, Rathkopf D, Smith MR, Efstathiou E, Attard G, Olmos D, et al. Niraparib and abiraterone acetate for metastatic castration-resistant prostate cancer. J Clin Oncol. 2023;41:3339–51.
Triggiani L, Mazzola R, Magrini SM, Ingrosso G, Borghetti P, Trippa F, et al. Metastasis-directed stereotactic radiotherapy for oligoprogressive castration-resistant prostate cancer: a multicenter study. World J Urol. 2019;37:2631–7.
Graham LS, Montgomery B, Cheng HH, Yu EY, Nelson PS, Pritchard C, et al. Mismatch repair deficiency in metastatic prostate cancer: response to PD-1 blockade and standard therapies. PLoS ONE. 2020;15:e0233260.
Fizazi K, Foulon S, Carles J, Roubaud G, McDermott R, Flechon A, et al. Abiraterone plus prednisone added to androgen deprivation therapy and docetaxel in de novo metastatic castration-sensitive prostate cancer (PEACE-1): a multicentre, open-label, randomised, phase 3 study with a 2 x 2 factorial design. Lancet. 2022;399:1695–707.
Badrising S, van der Noort V, van Oort IM, van den Berg HP, Los M, Hamberg P, et al. Clinical activity and tolerability of enzalutamide (MDV3100) in patients with metastatic, castration-resistant prostate cancer who progress after docetaxel and abiraterone treatment. Cancer. 2014;120:968–75.
Noonan KL, North S, Bitting RL, Armstrong AJ, Ellard SL, Chi KN. Clinical activity of abiraterone acetate in patients with metastatic castration-resistant prostate cancer progressing after enzalutamide. Ann Oncol. 2013;24:1802–7.
Dodkins J, Nossiter J, Cook A, Payne H, Clarke N, van der Meulen J, et al. Does research from clinical trials in metastatic hormone-sensitive prostate cancer treatment translate into access to treatments for patients in the “real world”? a systematic review. Eur Urol Oncol. 2024;7:14–24.
Shore ND, Laliberté F, Ionescu-Ittu R, Yang L, Mahendran M, Lejeune D, et al. Real-world treatment patterns and overall survival of patients with metastatic castration-resistant prostate cancer in the US prior to PARP inhibitors. Adv Ther. 2021;38:4520–40.
Basch E, Loblaw DA, Oliver TK, Carducci M, Chen RC, Frame JN, et al. Systemic therapy in men with metastatic castration-resistant prostate cancer: American Society of Clinical Oncology and Cancer Care Ontario Clinical Practice Guideline. J Clin Oncol. 2014;32:3436–48.
Lowrance W, Dreicer R, Jarrard DF, Scarpato KR, Kim SK, Kirkby E, et al. Updates to advanced prostate cancer: AUA/SUO guidelines (2023). J Urol. 2023;209:1082–90.
Fizazi K, Gillessen S. Updated treatment recommendations for prostate cancer from the ESMO Clinical Practice Guideline considering treatment intensification and use of novel systemic agents. Ann Oncol. 2023;34:557–63.
European Association of Urology: Prostate Cancer. 2024. https://uroweb.org/guidelines/prostate-cancer.
Dror MC, Chi KN, Khalaf DJ. Finding the optimal treatment sequence in metastatic castration-resistant prostate cancer-a narrative review. Transl Androl Urol. 2021;10:3931–45.
Lavaud P, Gravis G, Foulon S, Joly F, Oudard S, Priou F, et al. Anticancer activity and tolerance of treatments received beyond progression in men treated upfront with androgen deprivation therapy with or without docetaxel for metastatic castration-naïve prostate cancer in the GETUG-AFU 15 phase 3 trial. Eur Urol. 2018;73:696–703.
Halabi S, Kelly WK, Ma H, Zhou H, Solomon NC, Fizazi K, et al. Meta-analysis evaluating the impact of site of metastasis on overall survival in men with castration-resistant prostate cancer. J Clin Oncol. 2016;34:1652–9.
Pond GR, Sonpavde G, de Wit R, Eisenberger MA, Tannock IF, Armstrong AJ. The prognostic importance of metastatic site in men with metastatic castration-resistant prostate cancer. Eur Urol. 2014;65:3–6.
Annala M, Fu S, Bacon JVW, Sipola J, Iqbal N, Ferrario C, et al. Cabazitaxel versus abiraterone or enzalutamide in poor prognosis metastatic castration-resistant prostate cancer: a multicentre, randomised, open-label, phase II trial. Ann Oncol. 2021;32:896–905.
Deek MP, Taparra K, Phillips R, Velho PI, Gao RW, Deville C, et al. Metastasis-directed therapy prolongs efficacy of systemic therapy and improves clinical outcomes in oligoprogressive castration-resistant prostate cancer. Eur Urol Oncol. 2021;4:447–55.
Patel P, Dreibe S, Attard G, Cole A, Frew J, Guevara J, et al. Primary endpoint analysis of TRAP, first prospective Phase II trial of SBRT to oligoprogressive disease prostate cancer (NCT03644303). ESTRO:poster 3381, 2024.
Armstrong AJ, Lin P, Higano CS, Sternberg CN, Sonpavde G, Tombal B, et al. Development and validation of a prognostic model for overall survival in chemotherapy-naïve men with metastatic castration-resistant prostate cancer. Ann Oncol. 2018;29:2200–7.
Chi KN, Kheoh T, Ryan CJ, Molina A, Bellmunt J, Vogelzang NJ, et al. A prognostic index model for predicting overall survival in patients with metastatic castration-resistant prostate cancer treated with abiraterone acetate after docetaxel. Ann Oncol. 2016;27:454–60.
Halabi S, Yang Q, Roy A, Luo B, Araujo JC, Logothetis C, et al. External validation of a prognostic model of overall survival in men with chemotherapy-naive metastatic castration-resistant prostate cancer. J Clin Oncol. 2023;41:2736–46.
Pritchard CC, Mateo J, Walsh MF, De Sarkar N, Abida W, Beltran H, et al. Inherited DNA-repair gene mutations in men with metastatic prostate cancer. N Engl J Med. 2016;375:443–53.
Robinson D, Van Allen EM, Wu YM, Schultz N, Lonigro RJ, Mosquera JM, et al. Integrative clinical genomics of advanced prostate cancer. Cell. 2015;161:1215–28.
Abida W, Armenia J, Gopalan A, Brennan R, Walsh M, Barron D, et al. Prospective genomic profiling of prostate cancer across disease states reveals germline and somatic alterations that may affect clinical decision making. JCO Precis Oncol. 2017;2017:PO.17.00029.
Shore N, Ionescu-Ittu R, Yang L, Laliberte F, Mahendran M, Lejeune D, et al. Real-world genetic testing patterns in metastatic castration-resistant prostate cancer. Future Oncol. 2021;17:2907–21.
Ionescu F, Zhang J, Wang L. Clinical applications of liquid biopsy in prostate cancer: from screening to predictive biomarker. Cancers (Basel). 2022;14:1728.
Jensen K, Konnick EQ, Schweizer MT, Sokolova AO, Grivas P, Cheng HH, et al. Association of clonal hematopoiesis in DNA repair genes with prostate cancer plasma cell-free DNA testing interference. JAMA Oncol. 2021;7:107–10.
Fizazi K, Piulats JM, Reaume MN, Ostler P, McDermott R, Gingerich JR, et al. Rucaparib or physician’s choice in metastatic prostate cancer. N Engl J Med. 2023;388:719–32.
de Bono J, Mateo J, Fizazi K, Saad F, Shore N, Sandhu S, et al. Olaparib for metastatic castration-resistant prostate cancer. N Engl J Med. 2020;382:2091–102.
Li L, Karanika S, Yang G, Wang J, Park S, Broom BM, et al. Androgen receptor inhibitor-induced “BRCAness” and PARP inhibition are synthetically lethal for castration-resistant prostate cancer. Sci Signal. 2017;10:eaam7479.
Clarke NW, Armstrong AJ, Thiery-Vuillemin A, Mototsugu O, Shore N, Loredo E, et al. Abiraterone and olaparib for metastatic castration-resistant prostate cancer. NEJM Evidence. 2022;1:EVIDoa2200043.
AstraZeneca: Update on FDA Advisory Committee vote on Lynparza plus abiraterone for metastatic castration-resistant prostate cancer. 2023. https://www.astrazeneca.com/media-centre/press-releases/2023/odac-vote-on-lynparza-combo-in-prostate-cancer.html.
Fizazi K, Azad A, Matsubara N, Carles J, Fay AP, De Giorgi U, et al. TALAPRO-2: Phase 3 study of talazoparib (TALA) + enzalutamide (ENZA) versus placebo (PBO) + ENZA as first-line (1L) treatment for patients (pts) with metastatic castration-resistant prostate cancer (mCRPC) harboring homologous recombination repair (HRR) gene alterations. J Clin Oncol. 2023;41:5004.
Chi KNN, Castro E, Attard G, Smith MR, Sandhu SK, Efstathiou E, et al. LBA85 Niraparib (NIRA) with abiraterone acetate plus prednisone (AAP) as first-line (1L) therapy in patients (pts) with metastatic castration-resistant prostate cancer (mCRPC) and homologous recombination repair (HRR) gene alterations: three-year update and final analysis (FA) of MAGNITUDE. Ann Oncol. 2023;34:S1326.
Pfizer: TALZENNA (talazoparib) capsules, for oral use: summary of product characteristics. 2019. https://www.ema.europa.eu/en/documents/product-information/talzenna-epar-product-information_en.pdf.
AstraZeneca: LYNPARZA (olaparib) tablets, for oral use: summary of product characteristics. 2015. https://www.ema.europa.eu/en/documents/product-information/lynparza-epar-product-information_en.pdf.
Fallah J, Xu J, Weinstock C, Gao X, Heiss BL, Maguire WF, et al. Efficacy of poly(ADP-ribose) polymerase inhibitors by individual genes in homologous recombination repair gene-mutated metastatic castration-resistant prostate cancer: a US Food and Drug Administration pooled analysis. J Clin Oncol. 2024;42:1687–98.
Nava Rodrigues D, Rescigno P, Liu D, Yuan W, Carreira S, Lambros MB, et al. Immunogenomic analyses associate immunological alterations with mismatch repair defects in prostate cancer. J Clin Investig. 2018;128:4441–53.
Abida W, Cheng ML, Armenia J, Middha S, Autio KA, Vargas HA, et al. Analysis of the prevalence of microsatellite instability in prostate cancer and response to immune checkpoint blockade. JAMA Oncol. 2019;5:471–8.
Food and Drug Administration: FDA grants accelerated approval to pembrolizumab for first tissue/site agnostic indication. 2017. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-pembrolizumab-first-tissuesite-agnostic-indication.
GlaxoSmithKline: JEMPERLI (dostarlimab-gxly) injection, for intravenous use: summary of product characteristics. 2021. https://gskpro.com/content/dam/global/hcpportal/en_US/Prescribing_Information/Jemperli/pdf/JEMPERLI-PI-MG.PDF.
Hofman MS, Emmett L, Sandhu S, Iravani A, Joshua AM, Goh JC, et al. [(177)Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet. 2021;397:797–804.
Kuo P, Hesterman J, Rahbar K, Kendi AT, Wei XX, Fang B, et al. [68Ga]Ga-PSMA-11 PET baseline imaging as a prognostic tool for clinical outcomes to [177Lu]Lu-PSMA-617 in patients with mCRPC: A VISION substudy. J Clin Oncol. 2022;40:5002.
Hofman MS, Emmett L, Sandhu S, Iravani A, Buteau JP, Joshua AM, et al. Overall survival with [177Lu]Lu-PSMA-617 versus cabazitaxel in metastatic castration-resistant prostate cancer (TheraP): secondary outcomes of a randomised, open-label, phase 2 trial. Lancet Oncol. 2023;25:99–107.
Food and Drug Administration: FDA approves larotrectinib for solid tumors with NTRK gene fusions. 2018. https://www.fda.gov/drugs/fda-approves-larotrectinib-solid-tumors-ntrk-gene-fusions.
Food and Drug Administration: FDA approves entrectinib for NTRK solid tumors and ROS-1 NSCLC. 2019. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-entrectinib-ntrk-solid-tumors-and-ros-1-nsclc,
Food and Drug Administration: FDA grants accelerated approval to dabrafenib in combination with trametinib for unresectable or metastatic solid tumors with BRAF V600E mutation. 2022. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-dabrafenib-combination-trametinib-unresectable-or-metastatic-solid.
Food and Drug Administration: FDA approves selpercatinib for locally advanced or metastatic RET fusion-positive solid tumors. 2022. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-selpercatinib-locally-advanced-or-metastatic-ret-fusion-positive-solid-tumors.
Kellokumpu-Lehtinen PL, Harmenberg U, Joensuu T, McDermott R, Hervonen P, Ginman C, et al. 2-weekly versus 3-weekly docetaxel to treat castration-resistant advanced prostate cancer: a randomised, phase 3 trial. Lancet Oncol. 2013;14:117–24.
Oudard S, Ratta R, Voog E, Barthelemy P, Thiery-Vuillemin A, Bennamoun M, et al. Biweekly vs triweekly cabazitaxel in older patients with metastatic castration-resistant prostate cancer. JAMA Oncol. 2023;9:1629–38.
Satapathy S, Mittal BR, Sood A, Das CK, Mavuduru RS, Goyal S, et al. (177)Lu-PSMA-617 versus docetaxel in chemotherapy-naïve metastatic castration-resistant prostate cancer: a randomized, controlled, phase 2 non-inferiority trial. Eur J Nucl Med Mol Imaging. 2022;49:1754–64.
Sartor O, Castellano Gauna DE, Herrmann K, de Bono JS, Shore ND, Chi KNN, et al. LBA13 phase III trial of [177Lu]Lu-PSMA-617 in taxane-naive patients with metastatic castration-resistant prostate cancer (PSMAfore). Ann Oncol. 2023;34:S1324–S1325.
Novartis Pharmaceuticals Corporation: Press Release. Novartis Pluvicto™ shows clinically meaningful and highly statistically significant rPFS benefit in patients with PSMA-positive metastatic castration-resistant prostate cancer in the pre-taxane setting. 2023. https://www.novartis.com/news/media-releases/novartis-pluvictotm-shows-clinically-meaningful-and-highly-statistically-significant-rpfs-benefit-patients-psma-positive-metastatic-castration-resistant-prostate-cancer-pre-taxane-setting.
UroToday: Press Release. ASCO 2024: PSMAfore: HRQoL and pain in a phase 3 study of 177Lu-PSMA-617 in taxane-naïve patients with mCRPC. 2024. https://www.urotoday.com/conference-highlights/asco-2024/asco-2024-prostate-cancer/152475-asco-2024-psmafore-hrqol-and-pain-in-a-phase-3-study-of-177lu-psma-617-in-taxane-naive-patients-with-mcrpc.html.
Emmett L, Subramaniam S, Crumbaker M, Joshua AM, Weickhardt AJ, Lee ST, et al. LBA84 Enzalutamide and 177Lu-PSMA-617 in poor-risk, metastatic, castration-resistant prostate cancer (mCRPC): a randomised, phase II trial: ENZA-p (ANZUP 1901). Ann Oncol. 2023;34:S1325.
ClinicalTrials.gov: An international prospective open-label, randomized, phase III study comparing 177Lu-PSMA-617 in combination with SoC, versus SoC alone, in adult male patients with mHSPC (PSMAddition). NCT04720157. 2024. https://www.clinicaltrials.gov/ct2/show/NCT04720157.
Jang A, Kendi AT, Sartor O. Status of PSMA-targeted radioligand therapy in prostate cancer: current data and future trials. Ther Adv Med Oncol. 2023;15:17588359231157632.
ClinicalTrials.gov: Study of I-131-1095 radiotherapy in combination with enzalutamide in patients with metastatic castration-resistant prostate cancer who are chemotherapy naive and have progressed on abiraterone (ARROW). NCT03939689. 2023. https://classic.clinicaltrials.gov/ct2/show/NCT03939689.
Hansen AR, Probst S, Tutrone RF, Osman MM, Delpassand ES, Nordquist LT, et al. 1400P Efficacy and safety of 177Lu-PNT2002 prostate-specific membrane antigen (PSMA) therapy in metastatic castration resistant prostate cancer (mCRPC): initial results from SPLASH. Ann Oncol. 2022;33:S1185.
ClinicalTrials.gov: 177Lu-PSMA-I&T for metastatic castration-resistant prostate cancer. NCT05204927. 2024. https://www.clinicaltrials.gov/ct2/show/NCT05204927.
ClinicalTrials.gov: Pembrolizumab plus 177Lu-PSMA-617 in patients with castration resistant prostate cancer. NCT05766371. 2024. https://clinicaltrials.gov/ct2/show/NCT05766371.
ClinicalTrials.gov: Dose-escalation study of cabozantinib in combination with lutetium-177 (177Lu)-PSMA-617 in patients with metastatic castration-resistant prostate cancer (CaboLu). NCT05613894. 2024. https://clinicaltrials.gov/ct2/show/NCT05613894.
Berthold DR, Pond GR, Soban F, de Wit R, Eisenberger M, Tannock IF. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: updated survival in the TAX 327 study. J Clin Oncol. 2008;26:242–5.
Fendler WP, Weber M, Iravani A, Hofman MS, Calais J, Czernin J, et al. Prostate-specific membrane antigen ligand positron emission tomography in men with nonmetastatic castration-resistant prostate cancer. Clin Cancer Res. 2019;25:7448–54.
Dorff TB, Narayan V, Forman SJ, Zang PD, Fraietta JA, June CH, et al. Novel redirected t-cell immunotherapies for advanced prostate cancer. Clin Cancer Res. 2022;28:576–84.
Exelixis I: Press Release. Exelixis and Ipsen announce positive results from phase 3 CONTACT-02 pivotal trial evaluating cabozantinib in combination with atezolizumab in metastatic castration-resistant prostate cancer. 2023. https://ir.exelixis.com/news-releases/news-release-details/exelixis-and-ipsen-announce-positive-results-phase-3-contact-02.
Boysen G, Rodrigues DN, Rescigno P, Seed G, Dolling D, Riisnaes R, et al. SPOP-mutated/CHD1-deleted lethal prostate cancer and abiraterone sensitivity. Clin Cancer Res. 2018;24:5585–93.
Ferraldeschi R, Nava Rodrigues D, Riisnaes R, Miranda S, Figueiredo I, Rescigno P, et al. PTEN protein loss and clinical outcome from castration-resistant prostate cancer treated with abiraterone acetate. Eur Urol. 2015;67:795–802.
Beltran H, Hruszkewycz A, Scher HI, Hildesheim J, Isaacs J, Yu EY, et al. The role of lineage plasticity in prostate cancer therapy resistance. Clin Cancer Res. 2019;25:6916–24.
Akaza H, Procopio G, Pripatnanont C, Facchini G, Fava S, Wheatley D, et al. Metastatic castration-resistant prostate cancer previously treated with docetaxel-based chemotherapy: treatment patterns from the PROXIMA prospective registry. J Glob Oncol. 2018;4:1–12.
Ryan CJ, Smith MR, de Bono JS, Molina A, Logothetis CJ, de Souza P, et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N Engl J Med. 2012;368:138–48.
Fizazi K, Scher HI, Molina A, Logothetis CJ, Chi KN, Jones RJ, et al. Abiraterone acetate for treatment of metastatic castration-resistant prostate cancer: final overall survival analysis of the COU-AA-301 randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2012;13:983–92.
Ryan CJ, Smith MR, Fizazi K, Saad F, Mulders PF, Sternberg CN, et al. Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2015;16:152–60.
Beer, Armstrong TM, Rathkopf DE AJ, Loriot Y, Sternberg CN, Higano CS, et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424–33.
de Wit R, de Bono J, Sternberg CN, Fizazi K, Tombal B, Wulfing C, et al. Cabazitaxel versus abiraterone or enzalutamide in metastatic prostate cancer. N Engl J Med. 2019;381:2506–18.
Fizazi K, Azad AA, Matsubara N, Carles J, Fay AP, De Giorgi U, et al. First-line talazoparib with enzalutamide in HRR-deficient metastatic castration-resistant prostate cancer: the phase 3 TALAPRO-2 trial. Nat Med 2024;30:257–64.
Acknowledgements
Medical writing and editing assistance were provided by Greg Rowe, MSc, of Spark, a division of Prime, New York, USA, funded by Novartis Pharmaceuticals Corporation. Neither Novartis Pharmaceuticals Corporation nor Prime influenced the content of this manuscript, nor did the authors receive financial compensation for authorship.
Funding
Editorial support for this manuscript was provided by Spark, a division of Prime, New York, USA, funded by Novartis Pharmaceuticals Corporation.
Ethics declarations
Competing interests
HDM has no conflicts of interest to disclose. TD has received writing support from Pfizer, and has personal consulting or advising relationships with AstraZeneca, Bayer, Janssen Pharmaceuticals, and Sanofi. AM has a consulting relationship with Astellas, AstraZeneca, Bayer, Janssen Pharmaceuticals, Exelixis, Myovant, Merck, Novartis, Pfizer, Telix, and Sanofi. Research collaborations and/or support was received from Astellas, Bayer, Myovant, Pfizer, and Sanofi. OS has received grants or contracts from Advanced Accelerator Applications, Amgen, AstraZeneca, Bayer, Constellation, Endocyte, Invitae, Janssen Pharmaceuticals, Lantheus, Merck, Progenics Pharmaceuticals Inc., and Teneobio; has received consulting fees from Advanced Accelerator Applications, Astellas, AstraZeneca, Bayer, Blue Earth Diagnostics, Bavarian Nordic, Bristol Myers Squibb, Clarity Pharmaceuticals, Clovis, Constellation, Dendreon, EMD Serono, Fusion, Isotopen Technologien Meunchen, Janssen Pharmaceuticals, Myovant, Myriad, Noria Therapeutics, Novartis, Noxopharm, Progenics Pharmaceuticals, Inc., POINT Biopharma Global Inc., Pfizer, Sanofi, Teneobio, Telix, and Theragnostics; has participated on data safety monitoring boards or advisory boards for AstraZeneca, Janssen Pharmaceuticals, Pfizer, and Myovant; and holds stock or stock options in Clarity Pharmaceuticals, Noria Therapeutics, Eli Lilly, Clovis, GlaxoSmithKline, AbbVie, Cardinal Health, and United Health Group. NS has personal consulting or advising relationships with Astellas, Dendreon, Janssen Pharmaceuticals, Bayer, Myriad, MDxHealth, Tolmar, Molvant, Pfizer, AstraZeneca, Merck, Genentech, Pfizer, Urogen, Guardant, Myovant, AbbVie, Amgen, Bristol Myers Squibb, Boston Scientific, Clovis Oncology, Exact Imaging, FerGene, Foundation Medicine, GConcology, Invitae, Nymox, Propella, Sanofi Genzyme, Sesen Bio, Exact Sciences, Pacific Edge, Cold Genesys, Genesis Care, Alessa, Akido, Arquer, Asleris, Clarity, Clovis, Ferring, ImmunityBio, Incyte, Lantheus, Eli Lilly, Minomic, NGM, Nonagen, Novartis, Pacific Edge, Photocure, PlatformQ, Profound, Promaxo, Protara, Speciality Networks, Telix, Vaxllon, Vessi, FIZe Medical, Accord, ANTeV, Bioprotect, Aura Biosciences, CG Oncology, Palette Life, and PREVIEW; and holds a leadership position with Alessa and Photocure. AJA received research support (to Duke) from Astellas, Pfizer, Bayer, Janssen Pharmaceuticals, Dendreon, BMS, AstraZeneca, Merck, Forma, Celgene, Amgen, and Novartis; and has personal consulting or advising relationships with Astellas, Epic Sciences, Pfizer, Bayer, Janssen Pharmaceuticals, Dendreon, BMS, AstraZeneca, Merck, Forma, Celgene, Clovis, Exact Sciences, Myovant, Exelixis, GoodRx, and Novartis.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
McManus, H.D., Dorff, T., Morgans, A.K. et al. Navigating therapeutic sequencing in the metastatic castration-resistant prostate cancer patient journey. Prostate Cancer Prostatic Dis (2024). https://doi.org/10.1038/s41391-024-00906-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41391-024-00906-z
Subjects


Comments
Post a Comment